Modules¶
MCRA is a modular system. The diagram of Figure 19 shows the modules and their relations. Each module is associated with its own type of data, and is linked to one or more other modules. Note that not all details can be fully shown in the scheme, for details consult the table below, which specifies all relations between the modules in MCRA.
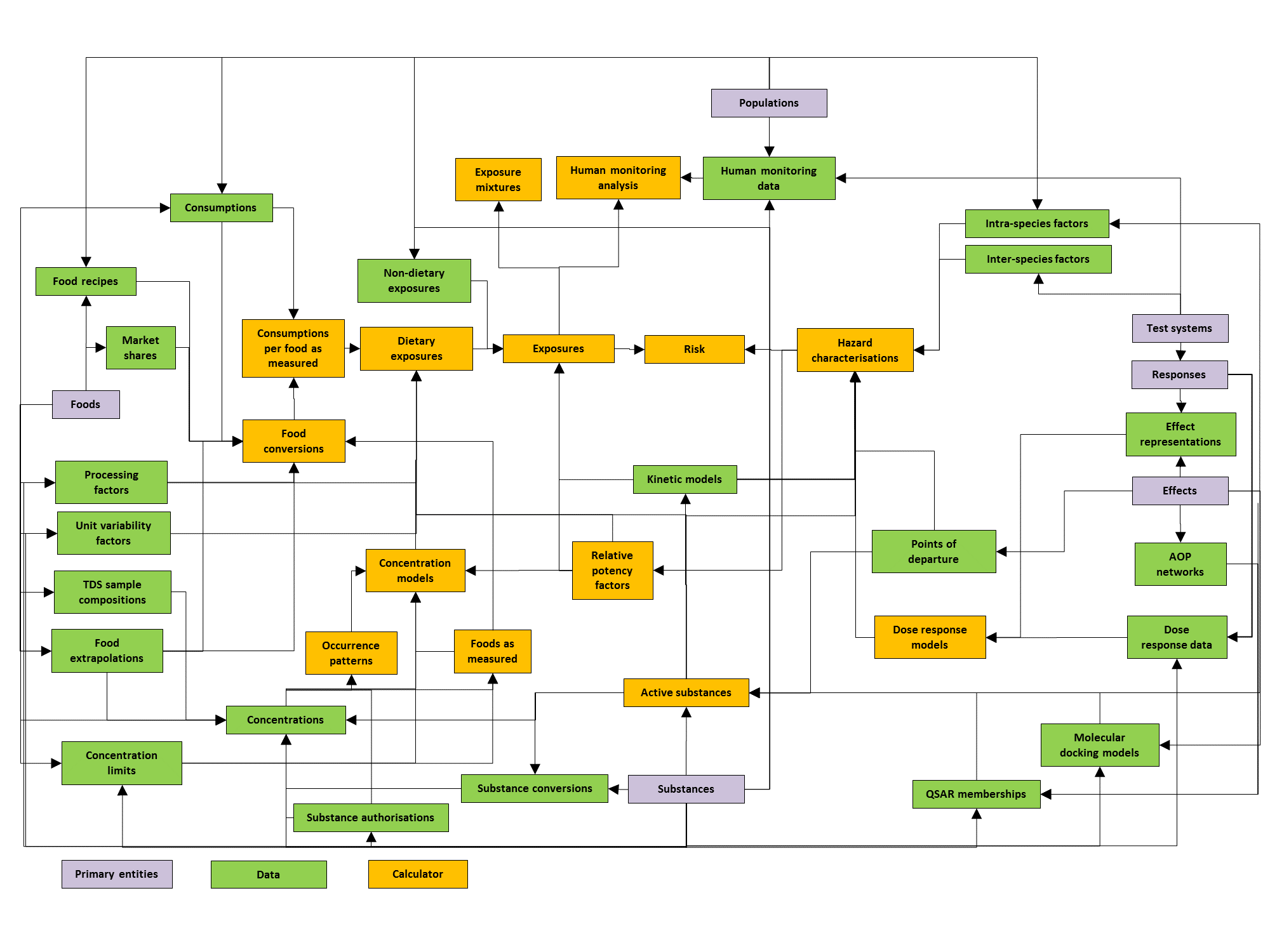
Figure 19 Diagram of the modular design of MCRA.¶
| Category | Module | Inputs | Used by | Description |
|---|---|---|---|---|
| Primary entities | Foods | Consumptions, Single value consumptions, Market shares, Food recipes, Concentrations, Concentration distributions, Single value concentrations, Processing factors, Unit variability factors, Occurrence patterns, Occurrence frequencies, Substance authorisations, Deterministic substance conversion factors, Concentration limits, Concentration models, Modelled foods, Focal food concentrations, Total diet study sample compositions, Food extrapolations, Food conversions, Consumptions by modelled food, High exposure food-substance combinations, Dietary exposures, Single value dietary exposures, Exposures, Exposure mixtures. | Foods are uniquely defined sources of dietary exposure to chemical substances. Foods may refer to 1) foods as eaten, foods as coded in food consumption data (e.g. pizza); 2) modelled foods, foods as coded in concentration data (e.g. wheat, tomato); 3) any other type of food (e.g. ingredients like flour, tomato sauce). | |
| Substances | Concentrations, Concentration distributions, Single value concentrations, Processing factors, Unit variability factors, Occurrence patterns, Occurrence frequencies, Substance authorisations, Substance conversions, Deterministic substance conversion factors, Concentration limits, Concentration models, Modelled foods, Focal food concentrations, Food conversions, Consumptions by modelled food, High exposure food-substance combinations, Dietary exposures, Single value dietary exposures, Non-dietary exposures, Exposures, Exposure mixtures, Human monitoring data, Human monitoring analysis, QSAR membership models, Molecular docking models, Kinetic models, Active substances, Relative potency factors, Hazard characterisations, Points of departure, Dose response models, Dose response data, Inter-species conversions, Intra species factors, Risks, Single value risks. | Substances are chemical entities that can refer to: 1) active substances such as investigated in toxicology; 2) measured substances such as defined in specific analytical methods. MCRA assessments can have one or more substances as the scope. When more than one substance is specified, there is an option to perform a cumulative assessment. In that case one of the substances has to be indicated as the index/reference substance, and results will be expressed in equivalents of the index substance. | ||
| Effects | Concentration models, High exposure food-substance combinations, Dietary exposures, Exposure mixtures, QSAR membership models, Molecular docking models, Active substances, Relative potency factors, Hazard characterisations, Points of departure, Effect representations, Inter-species conversions, Intra species factors, AOP networks, Risks, Single value risks. | Effects are biological or toxicological consequences for human health, that may result from chemical exposure and are the focus of hazard or risk assessment. | ||
| Populations | Consumptions, Single value consumptions, Consumptions by modelled food, Dietary exposures, Single value dietary exposures, Non-dietary exposures, Exposures, Human monitoring analysis, Hazard characterisations, Risks, Single value risks. | Populations are groups of human individuals that are the scope of exposure or risk assessments. Optional descriptors of populations are location (e.g. a country), time period (start date, end date), age range (age min , age max) and gender. Example: the French population in 2005-2007 (= time period) of women (= gender) of child-bearing age 18-45 yr (= age range). | ||
| Test systems | Responses, Dose response models, Dose response data. | Test systems are biological or artificial systems used for assessing hazard in relation to chemical exposure from substances in varying doses. Test systems may refer to 1) in-vivo test systems (e.g. a rat 90-day study, a human biomonitoring study); 2) in-vitro test systems (e.g. HepaRG cells). | ||
| Responses | Test systems. | Dose response models, Dose response data, Effect representations. | Responses are measurable entities in test systems. Responses are used to represent effects (see effect representations) and their measured values are collected in dose response data. | |
| Consumption | Consumptions | Populations, Foods. | Food conversions, Consumptions by modelled food. | Consumptions data are the amounts of foods consumed on specific days by individuals in a food consumption survey. For acute exposure assessments, the interest is in a population of person-days, so one day per individual may be sufficient. For chronic exposure assessments, the interest is in a population of persons, so preferably two or more days per individual are needed. |
| Single value consumptions | Consumptions by modelled food. | Single value dietary exposures. | Single value consumption data are the single value amounts (Large Portion, Mean Consumption, p97.5Consumption) of modelled foods (foods-as-measured) consumed in a population. | |
| Market shares | Foods. | Food conversions. | Market shares data specify for a given food, percentages of more specific foods (subfoods, e.g. brands) representing their share in a market. Market shares are used when consumption data are available at a more generalised level than concentration data. | |
| Food recipes | Foods. | Food conversions. | Food recipes data specify the composition of specific foods (typically: foods-as-eaten) in terms of other foods (intermediate foods or foods-as-measured) by specifying proportions in the form of a percentage. | |
| Occurrence | Concentrations | Foods, Substances, Focal food concentrations, Food extrapolations, Substance conversions, Deterministic substance conversion factors, Relative potency factors, Substance authorisations, Active substances, Concentration limits. | Single value concentrations, Occurrence patterns, Concentration models, Modelled foods. | Concentrations data are analytical measurements of chemical substances occurring in food samples. In their simplest form, concentration data can just be used as provided by datasets. Optionally, concentrations data can be manipulated for active substances, extrapolated to other foods, and/or default values can be added for water. |
| Concentration distributions | Foods, Substances. | Concentration models, Dietary exposures. | Concentration distributions describe substance concentrations on foods in the form of summary statistics. | |
| Single value concentrations | Active substances, Concentrations, Concentration limits, Deterministic substance conversion factors. | Modelled foods, Single value dietary exposures. | Single value concentrations data are the single value estimates (High Residue, Maximum Residue Limit, Supervised Trials Median Residue) of residue concentrations on modelled foods. | |
| Processing factors | Foods, Substances. | Food conversions, Dietary exposures, Single value dietary exposures. | Processing factors are multiplication factors to derive the concentration in a processed food from the concentration in an unprocessed food and can be specified for identified processing types (e.g., cooking, washing, drying). Processing factors are primarily used in dietary exposure assessments to correct for the effect of processing on substance concentrations in dietary exposure calculations. | |
| Unit variability factors | Foods, Substances. | Dietary exposures, Single value dietary exposures. | Unit variability factors specify the variation in concentrations between single units of the same food, which have been put together in a mixture sample on which the concentration measurements have been made. Unit variability factors are used to account for the fact that concentration data often relate to composite samples, whereas an acute risk may result from single food units. | |
| Occurrence patterns | Substance authorisations, Active substances, Concentrations. | Occurrence frequencies, Dietary exposures. | Occurrence patterns (OPs) are the combinations (or mixtures) of substances that occur together on foods and the frequencies of these mixtures occurring per food, expressed in percentages. In the context of pesticides, occurrence patterns are associated with agricultural use percentages. Occurrence patterns are relevant to account for co-occurrence of active substances in exposed individuals. Occurrence patterns may be specified as data or modelled based on observed patterns of positive concentrations. | |
| Occurrence frequencies | Active substances, Occurrence patterns. | Concentration models, Single value dietary exposures. | Occurrence frequencies specify how often substances occur on foods. Frequencies are expressed as percentages. | |
| Substance authorisations | Foods, Substances. | Concentrations, Occurrence patterns, Concentration models. | Substance authorisations specify which food/substance combinations are authorised for (agricultural) use. If substance authorisations are used, then only the food/substance combinations that are specified in the data are assumed to be authorised and all other combinations are assumed to be not authorised. This information may, for instance, be used to determine whether concentration measurements below the LOR could be assumed true zeros. I.e., if a food/substance combinations is assumed to be unauthorised, then the LOR may be assumed to be a zero. | |
| Substance conversions | Substances, Active substances. | Concentrations. | Substance conversions specify how measured substances are converted to active substances, which are the substances assumed to cause health effects. In the pesticide legislation such measured substances and the substance conversion rules are known as residue definitions. | |
| Deterministic substance conversion factors | Substances, Foods. | Concentrations, Single value concentrations. | Deterministic substance conversion factors. | |
| Concentration limits | Foods, Substances. | Concentrations, Single value concentrations, Concentration models, Modelled foods. | Concentration limits specify (legal) limit values for substance concentrations on foods and are sometimes used as conservative values for concentration data. In the framework of pesticides the legal Maximum Residue Limit (MRL) is the best known example. | |
| Concentration models | Concentrations, Concentration limits, Modelled foods, Substance authorisations, Occurrence frequencies, Relative potency factors, Concentration distributions, Total diet study sample compositions. | High exposure food-substance combinations, Dietary exposures. | Concentration models are distributional models of substance concentrations on foods. They describe both the substance presence (yes/no, with no representing an absolute zero concentration) and the substance concentrations. Concentration models are specified per food/substance combination. | |
| Modelled foods | Concentrations, Single value concentrations, Concentration limits. | Concentration models, Food conversions. | Modelled foods are foods within the foods scope for which concentration data or MRLs of substances are available (or expected). | |
| Focal food concentrations | Foods, Substances. | Concentrations. | In some cases the attention in an assessment is on a specific food (focal food), against the background of other foods. Focal food concentrations are separate concentration data for one or more focal food commodities, that will take the place of any other concentration data for the focal food in the ordinary concentration data. | |
| Total diet study sample compositions | Foods. | Concentration models, Food conversions. | Total diet study sample compositions specify the composition of mixed food samples, such as used in a total diet study (TDS), in terms of their constituting foods. | |
| Food extrapolations | Foods. | Concentrations, Food conversions. | Food extrapolations data specify which foods (data rich foods) can be used to impute concentration data for other foods with insufficient data (data poor foods). | |
| Exposure | Food conversions | Consumptions, Modelled foods, Processing factors, Food recipes, Market shares, Food extrapolations, Total diet study sample compositions, Active substances. | Consumptions by modelled food, Dietary exposures. | Food conversions relate foods-as-eaten, as found in the consumption data, to modelled foods (foods-as-measured), which are the foods for which concentration data are available. A food-as-eaten can be linked to one, or multiple modelled foods using various conversion steps (e.g., using food recipes to translate a composite food into its ingredients). There are several types of conversion steps, and a conversion path may comprise multiple conversion steps between a food-as-eaten and a modelled food. |
| Consumptions by modelled food | Consumptions, Food conversions. | Single value consumptions, High exposure food-substance combinations, Dietary exposures. | Consumptions by modelled food are consumptions of individuals expressed on the level of the foods for which concentration data are available (i.e., the modelled-foods). These are calculated from consumptions of foods-as-eaten and food conversions that link the foods-as-eaten amounts to modelled-foods amounts. | |
| High exposure food-substance combinations | Consumptions by modelled food, Concentration models, Active substances, Relative potency factors. | Dietary exposures. | Identification of food-as-eaten/modelled food/substance combinations that have the highest expected contribution to exposure based on a simple screening model. | |
| Dietary exposures | Consumptions by modelled food, Concentration models, Processing factors, Unit variability factors, High exposure food-substance combinations, Active substances, Occurrence patterns, Relative potency factors, Food conversions, Concentration distributions. | Exposures. | Dietary exposures are the amounts of substances, expressed per kg bodyweight or per individual, to which individuals in a population are exposed from their diet per day. Depending on the exposure type, dietary exposures can be short-term/acute exposures and then contain exposures for individual-days, or they can be long-term/chronic exposures, in which case they represent the average exposure per day over an unspecified longer time period. | |
| Single value dietary exposures | Single value consumptions, Single value concentrations, Processing factors, Unit variability factors, Occurrence frequencies. | Single value risks. | Single value dietary exposures are based on the single value concentrations of substances, expressed per standard (kg) bodyweight and/or single value amounts of consumed modelled food. Depending on the exposure type, dietary exposures can be short-term/acute exposures. | |
| Non-dietary exposures | Populations, Substances, Active substances. | Exposures. | Non-dietary exposures are the amounts of substances to which individuals in a population are exposed via any of three non-dietary routes: dermal, inhalation or oral, per day. | |
| Exposures | Dietary exposures, Non-dietary exposures, Active substances, Relative potency factors, Kinetic models. | Exposure mixtures, Human monitoring analysis, Risks. | Exposures are amounts of substances, typically expressed per mass unit and per day, to which individuals in a population are exposed at a chosen target level. This target level may be external exposure (dietary exposure, expressed per unit body weight, or per person) or internal exposure (expressed per unit organ weight). Internal exposures may be aggregated from dietary and non-dietary exposures using either absorption factors or kinetic models to translate the external exposures to internal exposures. Exposures can be short-term/acute exposures and then contain exposures for individual-days, or they can be long-term/chronic exposures, in which case they represent the average exposure per day over an unspecified longer time period. | |
| Exposure mixtures | Exposures. | Exposure mixtures will select sets of co-occurring substances (one or more) that contribute most to the overall exposure patterns. | ||
| Human monitoring data | Substances. | Human monitoring analysis. | Human monitoring data quantify substance concentrations found in humans collected in human monitoring surveys. | |
| Human monitoring analysis | Human monitoring data, Exposures. | Human monitoring analysis compares observed human monitoring data with predictions made for the same population of individuals from dietary survey data, concentration data and (optionally) non-dietary exposure data. | ||
| In-silico | QSAR membership models | Substances, Effects, AOP networks. | Active substances. | QSAR membership models specify assessment group memberships for active substances related to a specific health effect (adverse outcome). Memberships should be derived externally from Quantitative Structure-Activity Relationship (QSAR) models. |
| Molecular docking models | Substances, Effects, AOP networks. | Active substances. | Molecular docking models specify binding energies for substances in specific molecular docking models related to a specific health effect (adverse outcome). | |
| Kinetic | Kinetic models | Substances, Active substances. | Exposures, Hazard characterisations. | Kinetic models convert exposures or hazard characterisations from one or more external routes or compartments to an internal (target) compartment. The reverse conversion from internal to external can also be made (reverse dosimetry). |
| Hazard | Active substances | AOP networks, Points of departure, Hazard characterisations, Molecular docking models, QSAR membership models. | Concentrations, Single value concentrations, Occurrence patterns, Occurrence frequencies, Substance conversions, Non-dietary exposures, Kinetic models, Relative potency factors, Hazard characterisations, Inter-species conversions, Intra species factors, Food conversions, High exposure food-substance combinations, Dietary exposures, Exposures. | Active substances are substances that may lead (P>0) to a specific health effect (adverse outcome). Active substances are specified directly as data or calculated from POD presence, QSAR models or Molecular docking models. Active substances can have an assessment group membership 1 (crisp), or values in the range (0,1] (probabilistic). |
| Relative potency factors | Active substances, AOP networks, Hazard characterisations. | Concentrations, Concentration models, High exposure food-substance combinations, Dietary exposures, Exposures. | Relative potency factors (RPFs) quantify potencies of substances with respect to a defined effect, relative to the potency of a chosen index substance. RPFs can be used to express combined exposures of multiple substances in terms of a the exposure value of the chosen index substance (i.e., in index substance equivalents). In MCRA, hazard characterisations, and therefore also RPFs are based on mass units (e.g., µg), and not on mol units. RPFs can be different for different levels of the human organism (external, internal, specific compartment). RPFs can be given as data or computed from hazard characterisations. RPFs can be specified with uncertainty. Computation from uncertain hazard characterisations allows to include correlations between uncertain RPFs which originate from using the same index substance. | |
| Hazard characterisations | AOP networks, Active substances, Points of departure, Dose response models, Effect representations, Inter-species conversions, Intra species factors, Kinetic models. | Active substances, Relative potency factors, Risks, Single value risks. | Hazard characterisations are reference exposure values for active substances at the chosen biological target level (external or internal). Hazard characterisations may be specified for specific effects or for the critical effect as defined in hazard characterisation. Hazard characterisations are specified as external values (e.g. human based guidance values, such as ADI or ARfD) or are based on points of departure, such as BMDs from dose-response models or externally specified points of departure (NOAEL, LOAEL, MDS). The computation may involve assessment factors, e.g. for inter-species conversion, intra-species variation or additional sources of uncertainty. The calculation may also use kinetic models or absorption factors to convert external doses to internal doses or vice versa. | |
| Points of departure | Substances, Effects, AOP networks. | Active substances, Hazard characterisations. | Externally specified points of departure can be used as an alternative to calculated BMDs from dose response models. Points of departure can be of various types, such as NOAEL, LOAEL or BMD. They can be used to construct the list of active substances, to derive relative potency factors, and to perform health impact assessments. | |
| Dose response models | Dose response data, Effect representations. | Hazard characterisations. | Dose response models are models fitted to dose response data and can be provided as data or calculated using a local or remote version of PROAST. The main results for hazard and risk assessment are benchmark doses (BMDs), related to a specified substance, response, optionally covariate value, and the benchmark response (BMR). | |
| Dose response data | Substances, Test systems, Responses. | Dose response models. | Dose response data are data on response values of test systems at specified doses of substances (or mixtures of substances) from dose response experiments. | |
| Effect representations | Effects, Responses, AOP networks. | Hazard characterisations, Dose response models. | Effect representations specify the responses that can be used to measure specified effects and which response levels, the benchmark response (BMR), define the hazard limits for the effects. | |
| Inter-species conversions | Substances, Effects, Active substances. | Hazard characterisations. | Inter-species conversions specify how to convert a hazard characterisation for a given species to a hazard characterisation for humans. In the simplest approach, this specifies a fixed inter-species factor. In a higher tier, this specifies a geometric mean (GM) and geometric standard deviation (GSD) for a lognormal uncertainty distribution of the interspecies factor. Inter-species conversion are specified per effect and can be general or substance-specific. | |
| Intra species factors | Substances, Effects, Active substances. | Hazard characterisations. | Intra-species factors specify how to convert a hazard characterisation from the average to a sensitive human individual. | |
| AOP networks | Effects. | QSAR membership models, Molecular docking models, Active substances, Relative potency factors, Hazard characterisations, Points of departure, Effect representations. | Effects are related to each other using the toxicological concept of adverse outcome pathways (AOPs) and adverse outcome pathway networks (see https://aopwiki.org). Adverse Outcome Pathway (AOP) Networks specify how biological events (effects) can lead to an adverse outcome (AO) in a qualitative way through relations of upstream and downstream key events (KEs), starting from molecular initiating events (MIEs). Using AOPs, the adverse outcome (AO), e.g., liver steatosis, is linked to key events (KEs), e.g., triglyceride accumulation in the liver, and to molecular initiating events (MIEs), e.g., PPAR-alpha receptor antagonism. In general, multiple AOPs may lead to the same AO, and therefore AOP networks can be identified. | |
| Risks | Risks | Exposures, Hazard characterisations. | Single value risks. | Risks (health impacts) are defined as a function of exposure and hazard characterisation at a chosen biological level (external or internal). Risk metrics are margins of exposure (MOE) or hazard indices (HI) or more generalised MOE or HI distributions. |
| Single value risks | Single value dietary exposures, Hazard characterisations, Risks. | Single value risks are risk estimates obtained from combining single value exposures with single value hazard characterisations or as a percentile from a risk distribution. |